Lead tetrafluoride 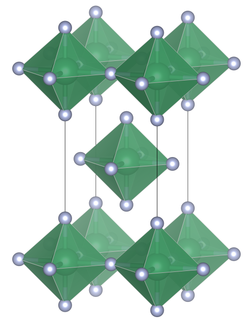
Unit cell of lead tetrafluoride |
| Names |
| IUPAC name Tetrafluoroplumbane |
| Other names Lead(IV) fluoride
Lead tetrafluoride
Tetrafluoridolead
Tetrafluoroplumbous anhydride |
| Identifiers |
CAS Number | - 7783-59-7
 Y Y
|
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.029.102  |
| EC Number | |
| | |
| UNII | - 5391K3G9PJ
 Y Y
|
| | - DTXSID40999040

|
InChI=1S/4FH.Pb/h4*1H;/q;;;;+4/p-4 Key: YAFKGUAJYKXPDI-UHFFFAOYSA-J
|
| |
| Properties |
Chemical formula | PbF4 |
| Molar mass | 283.194 g/mol [1] |
| Appearance | white to beige crystals [2] |
| Density | 6.7 g/cm3 [2] |
| Melting point | 600 °C (1,112 °F; 873 K)[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references |
Chemical compound
Lead tetrafluoride is a compound of lead and fluorine. The yellow solid (melting point 600 °C) is the only room-temperature stable tetrahalide of lead.[3] Lead tetrafluoride is isostructural with tin(IV) fluoride and contains planar layers of octahedrally coordinated lead, where the octahedra share four corners and there are two terminal, unshared, fluorine atoms trans to one another.[4]
References
- ^ "Lead(IV) fluoride | F4Pb - PubChem". Archived from the original on 2017-02-22. Retrieved 2017-02-22.
- ^ a b c "Lead Tetrafluoride | 7783-59-7".
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 375–376, 381–382. ISBN 978-0-08-037941-8.
- ^ Inorganic Chemistry [Paperback],2d Edition, Housecroft, Sharpe,2004, Pearson Education ISBN 0130399132, ISBN 978-0130399137
Lead compounds
| Pb(II) | - Pb(BiO3)2
- PbBr2
- Pb(C5H5)2
- Pb(C2H3O2)2
- PbC2O4
- PbC32H16N8
- PbCl2
- Pb(ClO4)2
- PbCO3
- PbCrO4
- PbF2
- PbHAsO4
- PbI2
- Pb(C
11H
23COO)
2 - Pb(NO3)2
- Pb(N3)2
- PbO
- Pb(OH)2
- PbPo
- PbP7
- Pb3(PO4)2
- PbS
- Pb(SCN)2
- PbSe
- PbSO4
- PbSeO4
- PbTe
- PbTiO3
- PbGeO3
- C
36H
70PbO
4 - plumbite
- PbC2 (hypothetical)
|
|---|
| Pb(II,IV) | |
|---|
| Pb(IV) | - Pb(C2H3O2)4
- PbCl4
- PbF4
- PbH4
- PbO2
- PbS2
- plumbate
- Pb(OH)4 (hypothetical)
|
|---|
Salts and covalent derivatives of the fluoride ion |
|---|
| HF | | | | ?HeF2 | | LiF | BeF2 | BF
BF3
B2F4
+BO3 | CF4
CxFy
+CO3 | NF3
FN3
N2F2
NF
N2F4
NF2
?NF5 | OF2
O2F2
OF
O3F2
O4F2
?OF4 | F2 | Ne | | NaF | MgF2 | AlF
AlF3 | SiF4 | P2F4
PF3
PF5 | S2F2
SF2
S2F4
SF3
SF4
S2F10
SF6
+SO4 | ClF
ClF3
ClF5 | ?ArF2
?ArF4 | | KF | CaF
CaF2 | | ScF3 | TiF2
TiF3
TiF4 | VF2
VF3
VF4
VF5 | CrF2
CrF3
CrF4
CrF5
?CrF6 | MnF2
MnF3
MnF4
?MnF5 | FeF2
FeF3
FeF4 | CoF2
CoF3
CoF4 | NiF2
NiF3
NiF4 | CuF
CuF2
?CuF3 | ZnF2 | GaF2
GaF3 | GeF2
GeF4 | AsF3
AsF5 | Se2F2
SeF4
SeF6
+SeO3 | BrF
BrF3
BrF5 | KrF2
?KrF4
?KrF6 | | RbF | SrF
SrF2 | | YF3 | ZrF2
ZrF3
ZrF4 | NbF4
NbF5 | MoF4
MoF5
MoF6 | TcF4
TcF
5
TcF6 | RuF3
RuF
4
RuF5
RuF6 | RhF3
RhF4
RhF5
RhF6 | PdF2
Pd[PdF6]
PdF4
?PdF6 | Ag2F
AgF
AgF2
AgF3 | CdF2 | InF
InF3 | SnF2
SnF4 | SbF3
SbF5 | TeF4
?Te2F10
TeF6
+TeO3 | IF
IF3
IF5
IF7
+IO3 | XeF2
XeF4
XeF6
?XeF8 | | CsF | BaF2 | | LuF3 | HfF4 | TaF5 | WF4
WF5
WF6 | ReF4
ReF5
ReF6
ReF7 | OsF4
OsF5
OsF6
?OsF
7
?OsF
8 | IrF2
IrF3
IrF4
IrF5
IrF6 | PtF2
Pt[PtF6]
PtF4
PtF5
PtF6 | AuF
AuF3
Au2F10
?AuF6
AuF5•F2 | Hg2F2
HgF2
?HgF4 | TlF
TlF3 | PbF2
PbF4 | BiF3
BiF5 | ?PoF2
PoF4
PoF6 | AtF
?AtF3
?AtF5 | RnF2
?RnF
4
?RnF
6 | | FrF | RaF2 | | LrF3 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | | | ↓ | | | | | LaF3 | CeF3
CeF4 | PrF3
PrF4 | NdF2
NdF3
NdF4 | PmF3 | SmF2
SmF3 | EuF2
EuF3 | GdF3 | TbF3
TbF4 | DyF2
DyF3
DyF4 | HoF3 | ErF3 | TmF2
TmF3 | YbF2
YbF3 | | AcF3 | ThF3
ThF4 | PaF4
PaF5 | UF3
UF4
UF5
UF6 | NpF3
NpF4
NpF5
NpF6 | PuF3
PuF4
PuF5
PuF6 | AmF2
AmF3
AmF4
?AmF6 | CmF3
CmF4
?CmF6 | BkF3
BkF
4 | CfF3
CfF4 | EsF3
EsF4
?EsF6 | Fm | Md | No | |
 | This inorganic compound–related article is a stub. You can help Wikipedia by expanding it. |













