Metsulfuron-methyl
Chemical compound
 | |
 | |
| Names | |
|---|---|
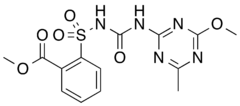
| IUPAC name 2-{[(4-methoxy-6-methyl-1,3,5-triazin-2-yl)amino]-oxomethyl]sulfamoyl}benzoic acid methyl ester | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| ECHA InfoCard | 100.129.351 |
| KEGG |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C14H15N5O6S |
| Molar mass | 381.36 g/mol |
| Density | 1.45 g/cm3 |
| Melting point | 163-166 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Metsulfuron-methyl is an organic compound classified as a sulfonylurea herbicide, which kills broadleaf weeds and some annual grasses.[1] It is a systemic compound with foliar and soil activity, that inhibits cell division in shoots and roots. It has residual activity in soils, allowing it to be used infrequently but requiring up to 22 months before planting certain crops (sunflowers, flax, corn, or safflower). It has very low toxicity to mammals, birds, fish, and insects but is a moderate eye irritant.[2]
References
- v
- t
- e
- acetochlor
- alachlor
- asulam
- benfluralin
- butachlor
- diethatyl
- diflufenican
- dimethenamid
- flamprop
- metazachlor
- metolachlor
- oryzalin
- pendimethalin
- pretilachlor
- propachlor
- propanil
- trifluralin
- aminopyralid
- chloramben
- clopyralid
- dicamba
- picloram
- pyrithiobac
- quinclorac
- quinmerac
- cacodylic acid
- copper arsenate
- DSMA
- MSMA
- flurochloridone
- isoxaflutole
- leptospermone
- mesotrione
- nitisinone
- sethoxydim
- sulcotrione
- bensulide
- bialaphos
- ethephon
- fosamine
- glufosinate
- glyphosate
- piperophos
| Auxins | |||||
|---|---|---|---|---|---|
| ACCase inhibitors |
|
| Nitrophenyl ethers | |
|---|---|
| Pyrimidinediones | |
| Triazolinones |
|
- dithiopyr
- fluroxypyr
- imazapyr
- thiazopyr
- triclopyr
| Photosystem I inhibitors |
|---|
| Photosystem II inhibitors |
|
|---|
| Photosystem II inhibitors |
|
|---|---|
| ALS inhibitors |
|
 | This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











