Vanadium(II) fluoride
 | |
| Names | |
|---|---|
| Other names Vanadium(II) fluoride | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
PubChem CID |
|
InChI
| |
| |
| Properties | |
Chemical formula | F2V |
| Molar mass | 88.9383 g·mol−1 |
| Appearance | blue crystals [1] |
Solubility in water | soluble in water, forms [V(H2O)6]2+[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Vanadium(II) fluoride is a fluoride of vanadium, with the chemical formula of VF2. It forms blue crystals.
Preparation
Vanadium(II) fluoride can be produced by the reduction of vanadium trifluoride by hydrogen in a hydrogen fluoride atmosphere at 1150 °C:[3]
- 2 VF3 + H2 → 2 VF2 + 2 HF
Properties
Physical properties
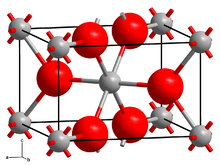
Vanadium(II) fluoride crystallizes in the tetragonal crystal system with space group P42/mnm (No. 136). Its lattice constants are a = 480.4 pm and c = 323.7 pm.[4]
Reactions
Vanadium(II) fluoride is a strong reducing agent that can reduce nitrogen to hydrazine in the presence of magnesium hydroxide.[2]
It dissolves in water to form [V(H2O)6]2+ ions.[2]
- V2+ + 6 H2O → [V(H2O)6]2+
References
- ^ "WebElements Periodic Table » Vanadium » vanadium difluoride". www.webelements.com. Retrieved 2023-09-16.
- ^ a b c Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, p. 1550, ISBN 0-12-352651-5
- ^ Lothar Kolditz: Anorganische Chemie Teil 2. VEB Deutscher Verlag der Wissenschaften, Berlin, 1980, S. 641.
- ^ J. W. Stout, W. O. J. Boo: Crystalline vanadium (II) fluoride, VF2. Preparation, structure, heat capacity from 5 to 300 K and magnetic ordering. In: The Journal of Chemical Physics. 71, 1, 1979, S. 1–8, doi:10.1063/1.438115.
- v
- t
- e
Vanadium compounds
- V(CO)6
- VF2
- VBr2
- VCl2
- VI2
- VO
- VS
- VSO4
- VBr3
- VCl3
- VF3
- VI3
- VN
- V2O3
- V2(SO4)3
- V2S3
| Organovanadium(III) compounds |
|---|
- VC
- VO2
- VOCl2
- V(S2)2
- VCl4
- VF4
| Organovanadium(IV) compounds | |
|---|---|
| Vanadyl(IV) compounds |
|
- V2O5
- VOCl3
- VOF3
- VO2F
- VF5
- VCl5
- NH4VO3
- VOPO4
- VO+2
| Vanadyl(V) compounds |
|
|---|











