Vanadium(V) chloride
 | |
 | |
| Names | |
|---|---|
| Other names vanadium pentachloride | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
PubChem CID |
|
InChI
| |
| |
| Properties | |
Chemical formula | Cl10V2 |
| Molar mass | 456.38 g·mol−1 |
| Appearance | black solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
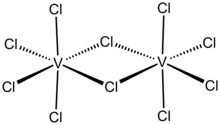
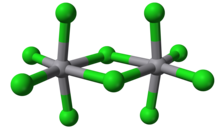
Vanadium(V) chloride is the inorganic compound with the formula VCl5. It is a black diamagnetic solid. The molecules adopt a bioctahedral structure similar to that of niobium(V) chloride.[1]
Preparation and reactions
Vanadium(V) chloride is prepared from the vanadium pentafluoride with excess boron trichloride:
- 2 VF5 + 10 BCl3 → [VCl5]2 + 10 BF2Cl
It is unstable at room temperature with respect to vanadium(IV) chloride:
- [VCl5]2 → 2 VCl4 + Cl2
In contrast, the heavier analogues NbCl5 and TaCl5 are stable and not particularly oxidizing.
References
- ^ Tamadon, Farhad; Seppelt, K. (2012). "The Elusive Halides VCl5, MoCl6, and ReCl6". Angewandte Chemie International Edition. 52 (2): 767–769. doi:10.1002/anie.201207552. PMID 23172658.
- v
- t
- e
Vanadium compounds
- V(CO)6
- VF2
- VBr2
- VCl2
- VI2
- VO
- VS
- VSO4
- VBr3
- VCl3
- VF3
- VI3
- VN
- V2O3
- V2(SO4)3
- V2S3
| Organovanadium(III) compounds |
|---|
- VC
- VO2
- VOCl2
- V(S2)2
- VCl4
- VF4
| Organovanadium(IV) compounds | |
|---|---|
| Vanadyl(IV) compounds |
|
- V2O5
- VOCl3
- VOF3
- VO2F
- VF5
- VCl5
- NH4VO3
- VOPO4
- VO+2
| Vanadyl(V) compounds |
|
|---|










